When Headlines Miss the Science: Inside Unity’s Senolytic Breakthrough
An insider's perspective reveals that while one of Unity’s Phase 2 trials missed its primary endpoint, their senolytic drug, UBX-1325, demonstrated compelling efficacy.
While offering one of the strongest validations to date of senescence as a therapeutic target.
When Unity Biotechnology announced results from their two Phase 2 trials — BEHOLD (phase 2) and ASPIRE (phase 2b) — the reaction was swift: headlines declared failure. But only one of those trials — ASPIRE — missed its primary endpoint. BEHOLD, a placebo-controlled study, met its pre-specified endpoint. While one technically missed its mark, both trials tell a compelling scientific story and delivered some of the most promising clinical data in the senolytic field to date.
Why two trials? BEHOLD and ASPIRE were designed to answer two distinct questions. BEHOLD asked: Does UBX-1325 work on its own?
ASPIRE asked: Can it outperform standard of care? Running both in parallel made strategic sense — isolating efficacy in one trial, while testing competitive positioning in the other.
And the results were revealing. BEHOLD confirmed UBX-1325’s standalone efficacy, with long-lasting improvements from a single injection. ASPIRE, though missing its primary endpoint, hinted at something even more profound: that over time, UBX-1325 might actually outperform standard of care, especially in patients who are in earlier stages of disease and have milder disease. Had ASPIRE run longer, it would likely have reached statistical significance. But in biotech, trial length is often a function of financial reality.
In an extended interview with Unity’s Chief Scientist, Mike Sapieha, PhD, we explored what the data actually shows, and why this may be a turning point for senescence-targeting therapeutics.
Understanding the Disease and the Mechanism
Unity’s drug candidate, UBX-1325, was developed for Diabetic Macular Edema (DME), the leading cause of vision loss in people with diabetes. The culprit? Senescent cells: damaged, non-dividing cells that linger in tissues and emit inflammatory signals, causing swelling and tissue dysfunction.
Whereas current treatments like Eylea suppress Vascular Endothelial Growth Factor to reduce retinal fluid, UBX-1325 takes a fundamentally different approach: eliminating the root cause (senescent cells) via BCL-xL inhibition, a member of the BCL-2 protein family that enables these pathiological senescent cells to avoid undergoing apoptosis.
"For the first time, we have a non-VEGF or non-steroid-based mechanism that showed clinically meaningful gains in visual acuity similar to VEGF in a controlled trial— at nearly all timepoints except one." – Mike Sapieha, PhD
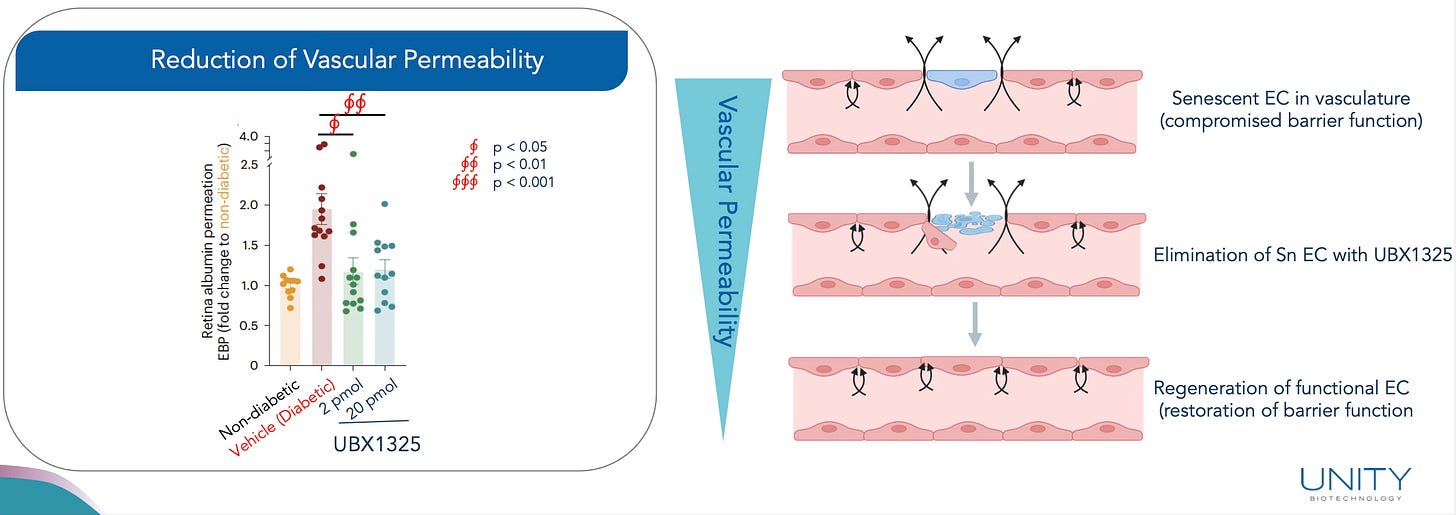
To understand why this matters, we can look at preclinical models. In a mouse model of diabetes, BCL-xL inhibition significantly reduced vascular leakage. As the image below shows, removing senescent endothelial cells helped restore barrier integrity and reduce inflammation-driven leakage.
Figure 1: BCL-xL inhibition (UBX1325) reduced retinal vascular permeability and restored endothelial function in diabetic mice. Source: Unity Corporate Deck
This isn’t just symptom management. It’s addressing the underlying pathology — by clearing the damaged cells that are causing the problem in the first place. And, more interestingly, not just clearing the damaged cells, but replacing them with youthful ones. Restoring integrity, reducing leakage, and ultimately preventing the buildup of fluid. (The fluid which Eylea is designed to remove.)
To put it simply: this is like a child spilling a glass of water on the table. Intuitively, the child starts wiping the floor — trying to clean up where the problem appears. But any parent would gently intervene: “Start with the table. Stop the source before mopping the floor.”
It’s a simple metaphor, but one that fits aging biology beautifully. Anti-VEGF therapies are wiping the floor. Senolytics go to the table and turn off the drip. Otherwise, we end up like Sisyphus — mopping forever, while the leak continues.
The BEHOLD Trial: The Forgotten Success
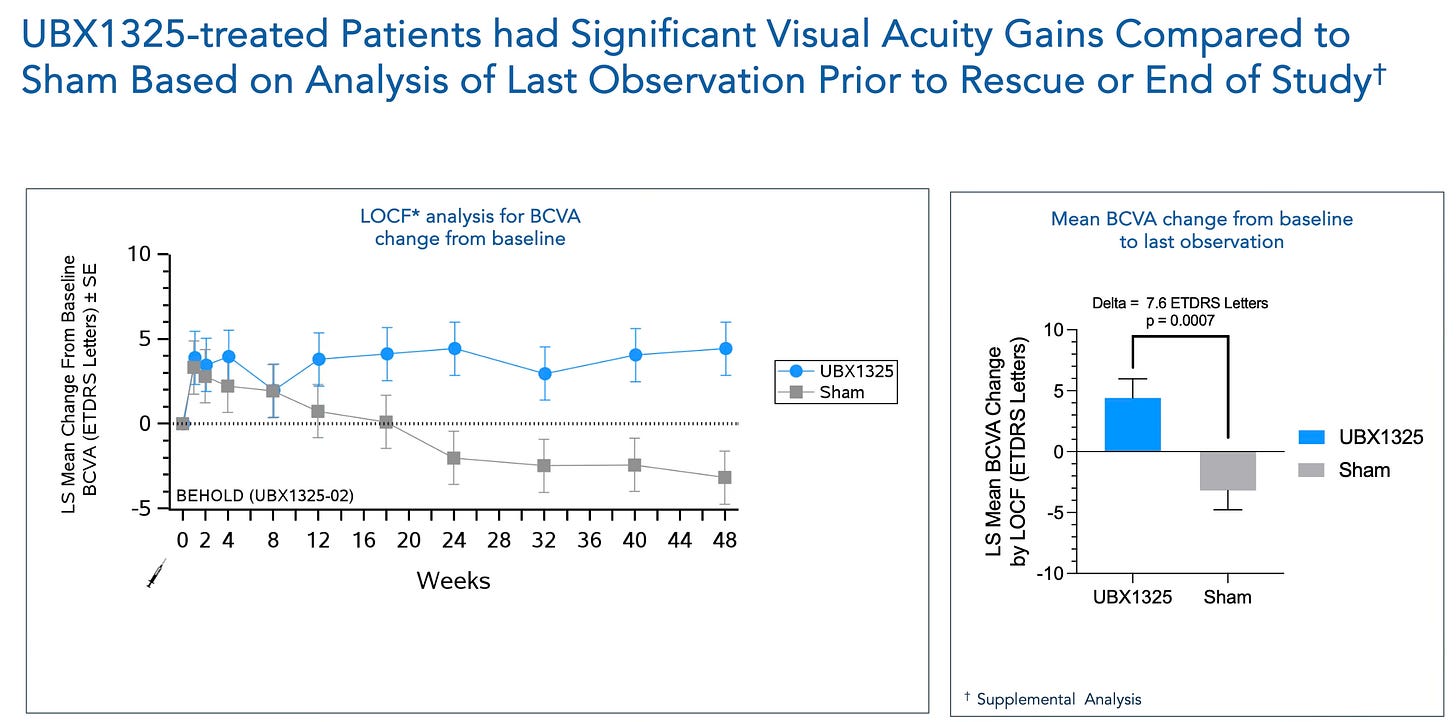
While headlines focused on missed endpoints, the BEHOLD trial met its pre-specified primary endpoint of safety and tolerability as well as its secondary endpoints of efficacy (BCVA improvement vs. sham), and showed strong and sustained visual acuity gains throughout the trial period.
At Week 48, patients on UBX-1325 retained vision gains significantly better than sham.
Over 30% improved their Diabetic Retinopathy Score.
~50% of patients didn’t require rescue injections over the course of 48 weeks.
Figure 2: Visual acuity improvements with UBX-1325 over 48 weeks vs. Sham. Source: Unity Corporate Deck, Slide 38.
Importantly, BEHOLD was a placebo-controlled trial — UBX-1325 was tested against sham injections, not aflibercept. This setup allowed Unity to isolate the effect of the senolytic alone, without the confounding influence of another active therapy. The results were notable.
Unity administered a single injection of UBX-1325 at baseline, with follow-up over 48 weeks. It was a bold design: a bet that one senolytic dose could deliver lasting clinical benefit. And it did — with sustained improvements in vision, reduced need for rescue injections, and signs of disease modification.
In contrast, the standard of care — anti-VEGF therapy — requires continuous dosing just to keep “mopping up” retinal fluid. It’s a classic symptom-management paradigm, and one that carries significant long-term economic burdens for healthcare systems already strained by the global rise in diabetes, aging populations, and falling birthrates. In the United States alone, the average sick span now exceeds 20 years. Without interventions that treat the root cause of chronic disease, the long-term economic and caregiving burden will become unsustainable.
The ASPIRE Trial: A Higher Bar, Different Stakes
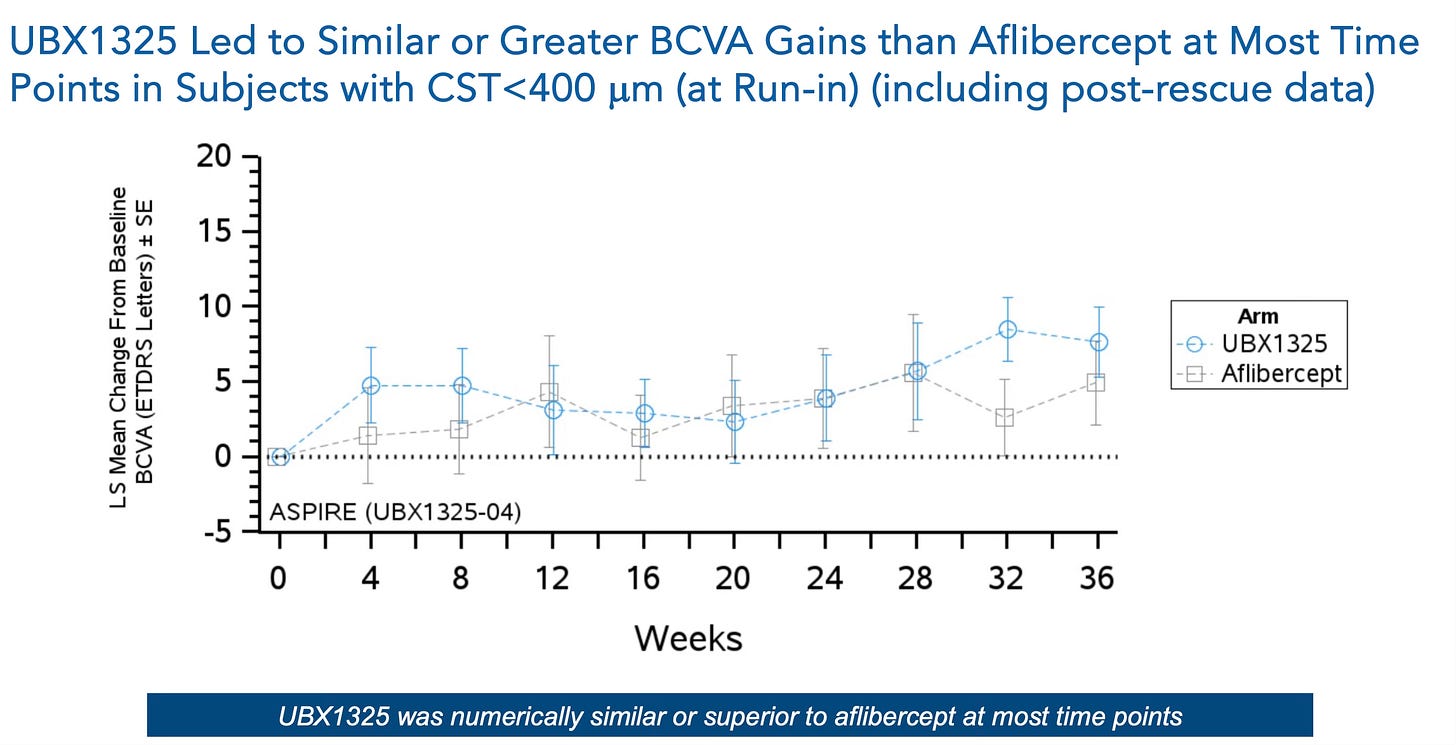
Unity also conducted the ASPIRE trial — a head-to-head study comparing UBX-1325 to aflibercept, the current standard of care. The goal: test whether a senolytic could compete directly with one of the most effective anti-VEGF agents on the market.
To do this, Unity matched aflibercept’s injection schedule, administering UBX-1325 every 8 weeks for three doses. The intent was clear: test efficacy, not convenience.
“We set our primary endpoint as change in BCVA on the average of weeks 20 and 24. Our statistical endpoint was set at >90% confidence interval and we met it at 88%, thus just missing the primary endpoint. When determining trial endpoints, we must consider what is clinically relevant and what our cash runway allows. While a 24-week endpoint is sound, it is certain that if our runway permitted, a primary read out at 36 or 48 weeks would have been more compelling.” — Mike Sapieha, PhD
Figure 3: UBX-1325 vs. Aflibercept visual acuity gains over time in ASPIRE. Source: Unity Corporate Deck
Importantly, this figure shows mixed results. Aflibercept performs strongly early on, a testament to its effectiveness in managing symptoms. But while aflibercept’s impact plateaued, UBX-1325 continued trending upward, suggesting the potential for rejuvenation of retinal tissue. So, as Mike stated, had the trial been extended beyond 24 weeks, this upward trajectory may have translated into clear superiority over the standard of care.
This aligns with the senolytic mechanism of action: rather than temporarily suppressing symptoms, senolytics remove the dysfunctional cells that drive disease — allowing healthier, younger cells to repopulate the tissue and repair damage.
Ultimately, the ASPIRE trial’s outcomes were mixed. The drug showed promising signs of efficacy, particularly in subgroups where the standard of care had non-responders, but did not meet all endpoints in the broader population.
What It All Means
Taken together, BEHOLD and ASPIRE tell a clear and compelling story:
BEHOLD delivered the first human validation of senolytics — proving that clearing senescent cells restores tissue function, improves vision, and reduces treatment burden. It showed that targeting BCL-xL is a viable strategy and established senolytics as a clinically relevant approach.
ASPIRE raised the bar by going head-to-head with the standard of care. In non-responders to anti-VEGF, UBX-1325 outperformed. In the broader population, it trended upward while aflibercept plateaued — suggesting that, with time, the senolytic could deliver even broader benefits.
Together, they don’t just validate a drug — they validate the modality. Senolytics are no longer theoretical. They work in humans. And they support a growing shift: from treating disease symptoms to targeting the underlying drivers of aging itself.
Why the Endpoint Was Set That Way
One of the most candid admissions in our conversation: the primary endpoint wasn’t selected because it was most clinically relevant — it was chosen because it fit Unity’s financial runway.
“Biotech has to design trials around clinically relevant timepoints and the cash available to reach them.” — Mike Sapieha, PhD
With capital tight and investors impatient, Unity had to structure its trial around constraints. That decision, while strategic, shaped the entire narrative around the trial's outcome.
This, unfortunately, is a common challenge for early-stage biotech companies. Not only must they navigate the scientific complexities of translating results from animal models to human trials, but they often do so under intense financial pressure. These constraints can impact trial design, endpoint selection, and even the perception of clinical success, irrespective of biological validity.
And in areas like senescence or deeptech, where the science is complex and mechanisms unfamiliar, perception often outweighs reality. Especially when companies are public and rely on generalist investors who may lack the tools to properly value novel therapeutic approaches. Headlines matter. Market sentiment reacts.
In that sense, Unity’s setback is arguably more of a commercial failure than a scientific one. Back in 2020, they faced disappointment with their knee osteoarthritis trial, so this was not their first stumble. Had this been a first-time failure, the market and investors might have responded with more patience. Instead, with a limited financial runway and past history in mind, investor sentiment turned quickly. Unity almost made it — but in this industry, 'almost' can be the difference between momentum and collapse.
That’s what ultimately led to a company-wide restructuring. Unity recently laid off its entire staff, including executive leadership. Unity is now set up a for a potential acquisition or partnership to push UBX-1325 into pivotal trials.
“We’ve validated the science. Now we need the right partner to take it forward.”
Unity is now positioned as a lean asset-ready company, awaiting a potential acquisition or partnership to push UBX-1325 into pivotal trials.
A New Treatment Paradigm: Vision vs. Fluid
Current treatments for DME focus on reducing fluid (edema) in the retina. But Unity’s drug showed a different benefit: restoration of vision.
“With UBX, you’re getting real visual gains. You’re stabilizing edema, but not drying the retina the same way as anti-VEGF. That suggests a combination therapy future.”
The most likely next step? A dual-therapy model: use anti-VEGF to dry the retina, then UBX-1325 to restore and maintain vision. This would address both symptom and cause.
The Future of Senolytics: From One-Size-Fits-All to Precision Medicine
To put Unity’s breakthrough into broader context, we asked an outside expert to weigh in: Marco Quarta, PhD, co-founder and CEO of Rubedo Life Sciences. As one of the industry leaders currently bringing second-generation senolytics into the clinic, Marco contributed this section to share where the field is heading — how Unity’s results signal a turning point in the broader evolution of senotherapeutics — not the end of a story, but the beginning of a more precise, layered, and ambitious chapter:
The early days of oncology taught us an important lesson: not all cancer cells are the same. What began as a blunt-force effort to eradicate tumors with non-specific agents evolved into a sophisticated arsenal of precision therapies — targeted inhibitors, immune checkpoint blockers, cell-based immunotherapies — tailored to specific mutations, cell types, and microenvironments. It took decades, billions of dollars, and more than a few high-profile failures. But today, oncology stands as one of medicine’s most advanced and hopeful frontiers.
Senotherapeutics is following a similar path.
At first, senescent cells were treated as a uniform target — damaged, inflammatory, and harmful. The mission was simple: eliminate them. Drugs like navitoclax (BCL-2/BCL-xL inhibitor), dasatinib + quercetin, and fisetin were broadly applied, aiming to purge these “zombie” cells and improve healthspan. But while those early trials showed tantalizing effects in both preclinical and small human studies — including in idiopathic pulmonary fibrosis and diabetic kidney disease — they also revealed limitations: low selectivity, off-target toxicity, and heterogeneous responses.
As the field matured, so did its understanding of senescence heterogeneity.
Thanks to single-cell multi-omics technologies — integrating single-cell RNA-seq, ATAC-seq, proteomics, and spatial transcriptomics — scientists began to see what bulk analyses obscured: senescence is not one state, but many. Senescent cells differ by tissue, stressor, cell lineage, and stage of senescence. Some are pro-inflammatory. Some are pro-fibrotic. Others play protective or regenerative roles, particularly in wound healing, embryogenesis, or immune modulation.
“We are no longer talking about one target. We’re talking about a taxonomy of senescence,” notes Marco Quarta, PhD. “Some cells should be removed. Others reprogrammed or preserved. That’s the future: selective and contextual senotherapeutics.”
This evolution is already underway.
Companies like Rubedo Life Sciences are using multi-omic profiling and AI-powered target discovery (e.g., ALEMBIC platform) to classify senescent subtypes, match them to diseases, and develop precision compounds. One such effort led to the discovery of novel druggable nodes in neuropathic conditions, now progressing toward translation.
Meanwhile, Unity Biotechnology’s UBX-1325 — though initially met with skepticism — demonstrated that even a single, well-timed senolytic dose can restore tissue function in human patients. That’s not just a milestone. It’s validation of the entire senolytic hypothesis: remove the harmful cells, and the tissue may heal itself.
The next generation of senotherapeutics is building on this foundation with:
Greater selectivity — using omics data to target only the pathogenic senescent subtypes.
Improved safety profiles — avoiding systemic toxicity and minimizing effects on beneficial cells.
Novel modalities — including antibody-drug conjugates (ADCs), senescence-targeted nanoparticles, immune-senolytics (engaging macrophages or NK cells), and epigenetic modulators that can reset or reprogram senescence rather than just kill.
And just like in oncology, combinatorial strategies are emerging. Some aim to pair senolytics with existing therapies (e.g., anti-VEGF, anti-fibrotics, or immunotherapies). Others propose “hit-and-run” approaches — brief, periodic interventions that clear senescent cells and then retreat, avoiding chronic exposure.
This modular, contextual approach allows the field to expand far beyond aging — into fibrosis, neurodegeneration, metabolic disease, osteoarthritis, even cancer relapse prevention. It is no longer a “longevity-only” play. It is a platform therapeutic space.
Unity’s story, while cautionary, is emblematic of the frontier: progress through trial, error, and bold experimentation. The setbacks are not signs of failure, but growing pains of a field maturing toward precision, personalization, and durability.
We are now entering a decade where the senolytic field will likely echo what oncology went through in the 1980s: a wave of new classifications, biomarkers, failure-driven refinements, and eventually — durable breakthroughs.
The age of “one drug to kill all senescent cells” is over. The era of precision senotherapeutics has begun.
And while it may take years to fully map the landscape and build the toolkit, the foundation is now clear: senescence is actionable and Second generation of Senolitics is already in the clinic, such as the RLS1496, a GPX4 modulator, currently tested in a basked clinical trial for I&I in chronic dermatological skin diseases and skin aging run by Rubedo Life Sciences. With the right tools, targets, and clinical strategies, we are no longer mopping the floor. We are turning off the tap.








Great share